Renowned Speakers

Mark-Cresswell
Lucedeon UK

Ulo Langel
Stockholm University Sweden

Reinhard Gabathuler
biOasis Technologies Inc. Canada

Pieter R Cullis
NanoMedicines Research Group Canada

Istvan Toth
The University of Queensland Australia Australia

Irach B Taraporewala
Sitara Pharmaceutical Consulting Group USA

Hiroshi Maeda
Sojo University Japan

Hajime Mori
Kyoto Institute of Technology Japan
Recommended Global Pharmaceutical Sciences Webinars & Conferences
Europe & UK
Asia Pacific & Middle East
Canada
Drug Delivery 2024
Drug Delivery Summit
May 16-17, 2024 | Rome, Italy
Welcome to the 18th World Drug Delivery Summit! We are delighted to invite you to this premier international gathering of professionals and experts in the field of drug delivery. This conference promises to be a platform for sharing knowledge, fostering innovation, and driving advancements in drug delivery technologies, all set against the backdrop of the historic and beautiful city of Rome, Italy.
|
Conference Name |
Place |
Date |
|
Rome, Italy |
May 16-17, 2024 |
About Conference
The Drug Delivery 2023 Summit is a distinguished event that brings together leading researchers, scientists, pharmaceutical professionals, clinicians, and industry experts from around the world. This two-day conference offers a unique opportunity to explore and discuss the latest developments, breakthroughs, and challenges in the realm of drug delivery.
Conference Highlights:
- Cutting-Edge Research: Engage with renowned experts as they present groundbreaking research findings in drug delivery systems, nanotechnology, biomaterials, and more.
- Interdisciplinary Collaboration: Foster collaboration between various disciplines, including pharmaceutical sciences, engineering, medicine, and chemistry, to accelerate drug delivery innovations.
- Keynote Speakers: Gain insights from keynote speakers who are pioneers in the field, offering their perspectives on the future of drug delivery.
- Interactive Workshops: Participate in interactive workshops and hands-on sessions to enhance your practical skills and knowledge.
- Exhibition Area: Explore a vibrant exhibition area featuring the latest advancements in drug delivery technologies, products, and services.
- Networking Opportunities: Connect with peers, researchers, and industry leaders, paving the way for potential collaborations and partnerships.
Why Attend:
- Stay Informed: Stay updated on the latest trends, challenges, and opportunities in drug delivery research and development.
- Connect: Network with professionals from academia, industry, and healthcare, fostering connections that can lead to future collaborations.
- Learn: Benefit from the insights of leading experts and gain valuable knowledge that can be applied to your work.
- Inspiration: Be inspired by cutting-edge research and innovative solutions that have the potential to improve patient outcomes.
Who Should Attend:
- Pharmaceutical Scientists
- Pharmacists
- Researchers and Academics
- Biomedical Engineers
- Clinical Pharmacologists
- Regulatory Affairs Specialists
- Materials Scientists
- Medical Practitioners
- Quality Assurance Professionals
- Marketing and Sales Specialists
Join us in Rome, Italy, on May 16-17, 2024, to be a part of this exceptional gathering. Be prepared to immerse yourself in the world of drug delivery, exchange ideas, and contribute to the future of healthcare. Together, let's shape the future of drug delivery and make a positive impact on patient care.
We look forward to welcoming you to the 18th World Drug Delivery Summit in the historic and enchanting city of Rome.
Session & Tracks
Session 1: "Recent Advances in Nanotechnology for Drug Delivery"
Nanotechnology has revolutionized drug delivery by enabling precise targeting and controlled release of therapeutic agents. This session will delve into the latest developments in this field, exploring nanoparticle-based drug carriers, their applications, and the challenges associated with nanomedicine.
Track A- Nanoparticle-based drug carriers: This subtopic will discuss the design and fabrication of nanoparticles as drug carriers. Attendees will learn about various types of nanoparticles, including liposomes, micelles, and polymeric nanoparticles, and how they enhance drug delivery.
Track B- Targeted drug delivery with nanoparticles: Nanoparticles can be engineered to target specific cells or tissues, improving drug efficacy while minimizing side effects. This subtopic will explore the strategies and success stories of targeted drug delivery using nanoparticles.
Track C- Clinical applications and successes: Participants will gain insights into real-world applications of nanotechnology in drug delivery. Case studies and clinical trials will be presented to highlight the achievements and potential of nanoparticle-based drug delivery systems.
Track D- Challenges and safety considerations in nanomedicine: While nanotechnology offers numerous benefits, it also raises safety concerns. This subtopic will address issues such as nanoparticle toxicity, regulatory hurdles, and strategies for ensuring the safe use of nanomedicine.
Session 2: "Personalized Medicine and Drug Delivery"
Personalized medicine has emerged as a transformative approach to healthcare, and it has a profound impact on drug delivery. This session will explore how pharmacogenomics, individualized dosing, and tailored formulations are shaping the future of drug delivery.
Track A- Pharmacogenomics and its role in drug delivery: Pharmacogenomics involves tailoring drug treatments based on an individual's genetic makeup. This subtopic will delve into how pharmacogenomics influences drug delivery system design and patient-specific therapies.
Track B- Individualized dosing and formulations: Attendees will learn about the development of personalized drug dosing regimens and formulations. Case studies and examples will showcase the practical implementation of individualized drug delivery.
Track C- Ethical and regulatory aspects of personalized medicine: Precision medicine raises ethical and regulatory questions related to genetic data, patient consent, and data protection. This subtopic will explore these challenges and propose solutions for responsible personalized drug delivery.
Session 3: "Innovations in Biomaterials for Drug Delivery"
Biomaterials play a crucial role in drug delivery systems, providing the scaffolding for controlled drug release. This session will highlight recent innovations in biomaterials, including smart biomaterials and bioresponsive drug carriers.
Track A: Smart biomaterials and responsive drug carriers: Attendees will explore the development of biomaterials that respond to specific environmental cues, allowing for precise drug release. Smart polymers, hydrogels, and responsive nanoparticles will be discussed.
Track B: Biodegradable materials in drug delivery: Biodegradable biomaterials are gaining popularity for their ability to break down in the body, eliminating the need for removal. This subtopic will showcase biodegradable drug delivery systems and their applications.
Track C- Biomaterials for tissue-specific targeting: Tissue-specific drug delivery is a critical aspect of precision medicine. This session will cover biomaterials designed to target specific tissues or organs, improving drug delivery accuracy.
Track D- Biomimetic drug delivery systems: Biomimetic biomaterials mimic natural biological processes, enhancing drug delivery efficiency. Attendees will learn about biomimetic drug carriers and their potential in personalized medicine.
Session 4: "Emerging Technologies in Drug Delivery"
Emerging technologies are shaping the future of drug delivery, enabling precise control and customization of therapeutic interventions. This session will explore cutting-edge technologies, including AI, 3D printing, and wearable drug delivery devices.
Track A- Artificial intelligence and machine learning in drug formulation: AI and machine learning are revolutionizing drug formulation and optimization. Attendees will discover how these technologies are being used to design personalized drug delivery systems.
Track B- 3D printing and personalized drug manufacturing: 3D printing offers a novel approach to manufacturing customized drug formulations and delivery devices. This subtopic will showcase the latest advancements in 3D-printed drug delivery.
Track C- Wearable drug delivery devices: Wearable devices are gaining prominence in drug delivery, offering continuous monitoring and precise administration. This session will delve into wearable drug delivery technologies and their clinical applications.
Track D- Remote monitoring of drug delivery: Attendees will explore how remote monitoring technologies are enhancing patient compliance and treatment outcomes in drug delivery. Case studies and real-world applications will be discussed.
Session 5: "Clinical Insights and Patient Outcomes"
This session focuses on the practical applications of drug delivery technologies in clinical practice. Attendees will gain insights into successful implementations, real-world evidence, and the impact of innovative drug delivery on patient outcomes.
Track A- Case studies of successful drug delivery in clinical practice: This subtopic will showcase real-world examples of drug delivery systems that have improved patient care and treatment outcomes across various medical specialties.
Track B- Real-world evidence and patient-reported outcomes: Attendees will learn about the importance of collecting and analyzing real-world evidence to validate the effectiveness and safety of drug delivery technologies. Patient-reported outcomes will also be discussed.
Track C- Quality of life improvements with innovative drug delivery: Precision drug delivery can significantly enhance patients' quality of life. Case studies and patient testimonials will illustrate how innovative drug delivery systems have made a positive impact.
Track D- Challenges in translating research to clinical impact: Despite promising research, translating drug delivery innovations into clinical practice can be challenging. This session will explore the hurdles and strategies for successful implementation.
Session 6: "Regulatory Pathways and Compliance in Drug Delivery"
Navigating the regulatory landscape is crucial for the development and approval of drug delivery systems. This session will provide insights into regulatory considerations, compliance, and ethical aspects.
Track A- FDA and EMA perspectives on drug delivery systems: Representatives from regulatory agencies will share their insights and perspectives on the evaluation and approval of drug delivery technologies, highlighting regulatory pathways and requirements.
Track B- Regulatory considerations for personalized medicine: Attendees will gain an understanding of the unique regulatory challenges and considerations associated with personalized drug delivery, including data protection and consent.
Track C- Data protection and patient consent in precision drug delivery: Ensuring patient data protection and informed consent are critical in personalized medicine. This subtopic will explore best practices and legal requirements.
Track D- Navigating the approval process for novel drug delivery technologies: This session will guide attendees through the complex process of gaining regulatory approval for innovative drug delivery systems, including clinical trial design and documentation.
Session 7: "Global Access and Societal Implications"
Achieving equitable global access to advanced drug delivery technologies is essential. This session will explore the economic, societal, and international dimensions of drug delivery innovations.
Track A- Equitable access to personalized drug delivery: Ensuring that personalized drug delivery is accessible to all populations, including underserved communities, will be discussed, along with strategies for reducing healthcare disparities.
Track B- Economic implications of precision medicine: Attendees will gain insights into the economic impact of precision medicine, including cost-effectiveness and economic modeling of personalized drug delivery.
Track C- International collaborations and initiatives in drug delivery: International efforts to promote collaborative research and access to drug delivery innovations will be highlighted, emphasizing the importance of global cooperation.
Track D- Addressing disparities in healthcare through advanced drug delivery: This session will explore how advanced drug delivery technologies can contribute to addressing healthcare disparities, both within and between countries.
Session 8: "Industry Innovations and Market Trends"
Industry plays a pivotal role in driving innovation and bringing drug delivery products to the market. This session will feature industry presentations, market trends, and investment opportunities in drug delivery.
Track A- Industry presentations on innovative drug delivery products: Representatives from leading pharmaceutical and biotechnology companies will showcase their latest drug delivery innovations, sharing insights into product development and market impact.
Track B- Market trends in drug delivery technology: Attendees will gain an understanding of current and future market trends in drug delivery technology, including growth sectors and investment opportunities.
Track C- Investment and funding opportunities in the drug delivery sector: This subtopic will provide insights into funding opportunities, partnerships, and investment strategies for startups and emerging companies in drug delivery.
Track D- Start-ups and entrepreneurship in drug delivery: This session will highlight successful startup ventures in the drug delivery sector, emphasizing the entrepreneurial spirit driving innovation.
Session 9: "Interactive Workshops and Demonstrations"
Interactive workshops and demonstrations provide attendees with hands-on experiences and practical knowledge in drug delivery. This session will offer engaging activities to enhance participants' skills and understanding.
Track A- Hands-on workshops on drug formulation: Workshops will guide attendees through the process of drug formulation, providing practical experience in designing drug delivery systems.
Track B- Demonstrations of cutting-edge drug delivery devices: Participants will have the opportunity to explore and interact with state-of-the-art drug delivery devices, gaining a firsthand understanding of their operation.
Track C- Simulation exercises on personalized dosing: Attendees will engage in simulation exercises to understand the principles of personalized dosing and its application in clinical practice.
Track D- Prototyping and testing of drug delivery prototypes: This subtopic will involve hands-on prototyping and testing of drug delivery prototypes, allowing participants to experiment with innovative designs.
Session 10: "Future Directions in Drug Delivery Research"
The final session will serve as a visionary forum for discussing the future of drug delivery research and innovation. Keynote speeches, panel discussions, and collaborative brainstorming sessions will provide attendees with insights into emerging challenges and opportunities.
Track A- Keynote speeches on the future of drug delivery: Renowned experts will deliver keynote speeches, offering their perspectives on the future direction of drug delivery, technological breakthroughs, and healthcare transformation.
Track B- TrackPanel discussions with visionary experts: Interactive panel discussions will bring together thought leaders to explore emerging trends, challenges, and visionary ideas in drug delivery research.
Track C- Collaborative brainstorming sessions on research priorities: Attendees will actively participate in brainstorming sessions to identify research priorities, innovative approaches, and strategies to address future healthcare needs.
Track D- Identifying emerging challenges and opportunities: This session will encourage attendees to identify and discuss emerging challenges and opportunities in drug delivery research, fostering a collaborative and forward-thinking environment.
These detailed notes provide an overview of the proposed sessions for the Drug Delivery Summit, covering a wide range of topics and ensuring that attendees receive valuable insights and knowledge across the field of drug delivery. Each session aims to engage participants, foster discussion, and promote innovation in drug delivery research and practice.
Market Analysis
Market Size
The global drug delivery systems market has shown significant growth. In 2022, it was valued at USD 39.55 billion, and it is projected to reach USD 78.76 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 9.1% during this period [1]. Additionally, the global drug delivery solutions market is expected to be valued at USD 107.5 billion in 2023 and is projected to grow at a CAGR of 6.8% between 2023 and 2033, reaching around USD 207.6 billion by 2033 [2]. These figures indicate a thriving market with substantial opportunities.
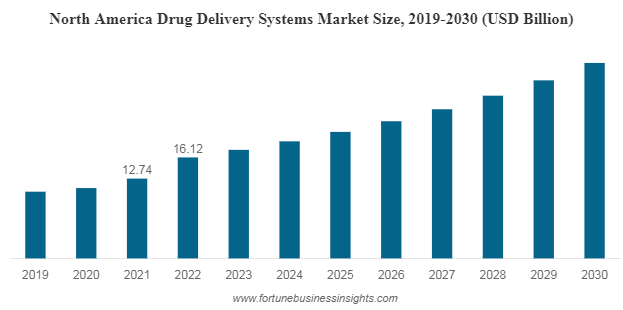
Trends
Several trends are shaping the drug delivery market:
- Biologics Delivery: There's a growing demand for effective delivery systems for biologic drugs, including gene therapies and monoclonal antibodies. Innovative delivery methods for these complex therapeutics are on the rise.
- Personalized Medicine: The trend towards personalized medicine is driving the development of targeted drug delivery systems tailored to individual patients' needs.
- Nanotechnology: Nanotechnology-based drug delivery systems are gaining traction due to their ability to improve drug solubility, bioavailability, and targeted delivery.
- Smart Drug Delivery: The integration of IoT and smart devices in drug delivery is enhancing patient adherence and monitoring, creating a more patient-centric approach.
Competition
The drug delivery market is highly competitive with key players including Pfizer, Johnson & Johnson, Novartis, and Roche. These companies invest heavily in research and development to introduce innovative drug delivery systems. Additionally, smaller firms and startups are focusing on niche areas and disruptive technologies.
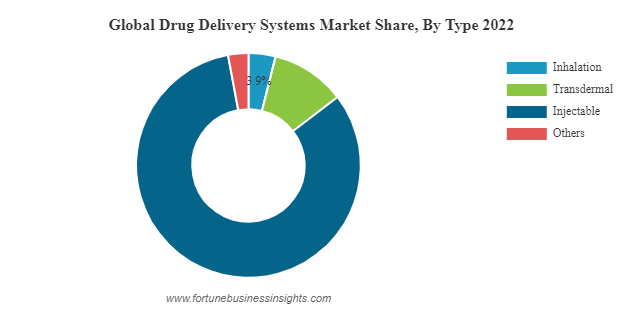
Future Projections
Based on current market trends, it is projected that the drug delivery market will continue to expand. Factors such as the aging population, increasing prevalence of chronic diseases, and advancements in drug delivery technologies will drive growth. The market is likely to witness further consolidation and collaborations as companies strive to gain a competitive edge.
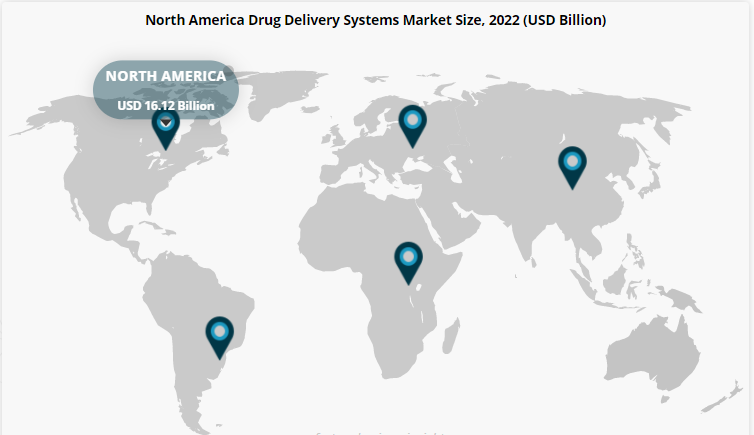
Projected Growth of the Drug Delivery Market
Conclusion
The drug delivery market is experiencing robust growth, driven by evolving trends and innovation. Stakeholders should focus on developing advanced delivery systems to cater to the rising demand for personalized and targeted therapies. Collaboration and strategic partnerships will be key in this competitive landscape.
For a detailed analysis and data sources, please refer to the cited references.
🌠Sources
Visa Process
For Visa Application process, please follow general steps as a guide. The application process is different for each country. It is your due diligence to check with the Italy Embassy within your country for the detailed procedure on visa application.
[ Step A: ]
In order for Drug Delivery 2024 organizers to provide an Invitation Letter for your visit, you must first register to attend Drug Delivery 2024. Once your registration is completed, you will receive a confirmation letter and an official receipt with your registration number/ID.
Click here to know more about the registration packages and fees.
[ Step B: ]
You will need an invitation letter to apply for visa. To request for one, applicant is required to provide his/her registration number/ID.
Processing and mailing time vary for each country so please allow 01-07 days for the visa invitation letter to reach your mailbox.
Please note that application for invitation letter is open to Conference Speakers, delegates, Students (Only registration & Package A/B) and Exhibitors.
The organizers of Drug Delivery 2024 reserve the rights to refuse any application if it is deemed suspicious of any illegal activities.
[ Step C: ]
You will also be required to fill up the visa application form and all other forms necessary as a part of the visa application process to enter Italy.
Other documents such as birth certificate, a copy of identification card and certificate of employment may be required from you. For the full list of documents required.
To request for further assistance regarding the visa application or requirements, kindly email to drugdelivery@annualforum.org , with your registration confirmation number/ID.
Note: Your visa application is dependent on the final decision of the Italy Embassy. Drug Delivery 2024 organizers cannot guarantee an approval/acceptance at any stage of the application process.
To Collaborate Scientific Professionals around the World
Conference Date May 16-17, 2024
Speaker Opportunity
Useful Links
Past Conference Report
Supported By
All accepted abstracts will be published in respective Conference Series International Journals.
Abstracts will be provided with Digital Object Identifier by















